 
With strong acids, for example, sulfuric and hydrochloric, aluminum reacts quite calmly, without ignition. It enters into the reaction as aluminum hydroxideĪnd as metallic aluminum or aluminum oxide: complex compounds which are actively used in the paper and textile industry. The silvery metal also reacts with complex chemical substances.įor example, with alkalis it forms aluminates. With other non-metals such as fluorine, sulfur, nitrogen, carbon etc. For example, with halogens, fluorine is the exception, aluminum can form aluminum iodide, chloride or bromide: An aluminothermic reaction using iron(III) oxideįor example, let us examine the reaction with chromium oxide:Īluminum easily reacts with simple substances. This feature of aluminum involves the interaction with oxides of other metals. 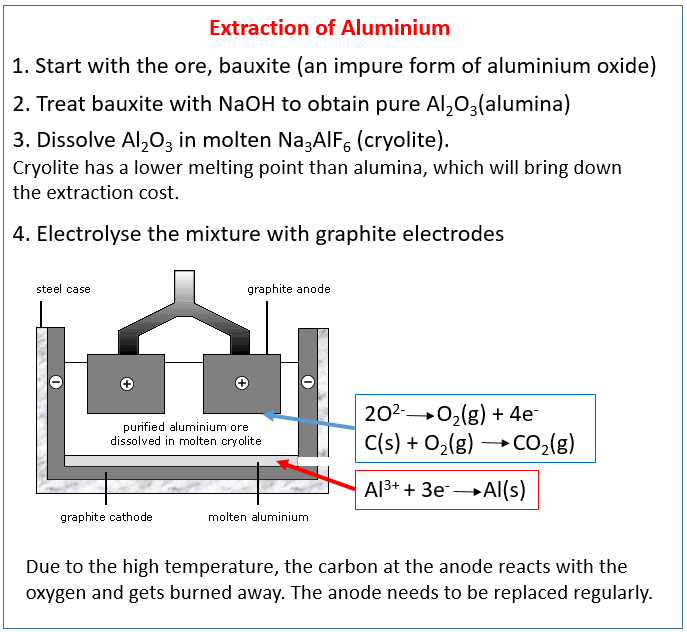
Thanks to its reducing properties, aluminum has found application in industry for obtaining other metals or non-metals, and this process is called aluminothermy. The oxide film of aluminum can be destroyed by a solution or alloy of alkalis, or with acids, and also with mercury chloride or oxide. But this film can be easily destroyed, so that the metal displays active reductive properties. If aluminum is left at room temperature, a durable protective layer of aluminum oxide Al₂O₃ forms on the metal, which protects it from corrosion.Īluminum practically does not react with oxidizers because of the oxide film that protects it. The melting point of aluminum is 660 ☌, and its boiling temperature is 2470 ☌. It is also very ductile, and can easily be turned into a thin sheet or foil. The physical properties of aluminumĪluminum is a silvery-white light metal, which can form alloys with the majority of metals, especially copper and magnesium, and also silicon. Metallic aluminum is safe, but its compounds can have a toxic effect on humans and animals, particularly aluminum chloride, acetate, and sulfate. Thanks to its malleability and lightness, and also its corrosion resistance, aluminum is a valuable metal of great importance in modern industry, from which such household items are made as kitchenware, and in industry: for example, it is widely used in aircraft and automobile construction.Īluminum is also one of the most inexpensive and economical materials, as it can be used endlessly, by melting down used aluminum cans and other objects. It is difficult to appreciate the value of this metal in its present form, as many items made of aluminum are used by people in everyday life. 
The industrial method involved the electrolysis of an alloy of cryolite, in which aluminum oxide was dissolved. But soon, in 1886, a method was developed by Charles Martin Hall and Paul Héroult for producing aluminum in industrial scale, which drastically reduced the cost of this metal, and allowed it to be used in the metallurgical industry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
